Structure of Alzheimer’s protein could explain why it’s toxic
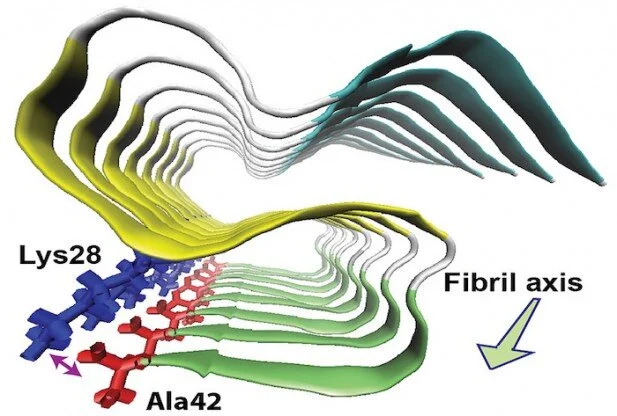
by Chuck Bednar for Red Orbit:In a potential breakthrough in the treatment of Alzheimer’s disease, researchers at the University of Illinois at Chicago have determined the molecular structure of one of the proteins found in the fibers of brain plaques linked to this and other neurodegenerative conditions.Yoshitaka Ishii, a professor of chemistry at the university and principal investigator of the study, and his colleagues found that amyloid beta-42, a molecule that is toxic to nerve cells, forms three flat structures known as beta sheets that layer over one another in an S-like pattern.Furthermore, as they report in the journal Nature Structural and Molecular Biology, there is an amino acid on the protein that forms a bond between positive and negative-charged parts of a molecule known as a salt bridge. This structure is significantly different from amyloid beta-40, a chemically similar and more abundant protein that has also been linked to Alzheimer’s.The salt bridge stabilizes the structure, and learning about the physical structure of the 42 amino acid-long form of amyloid beta (which experts believe may provoke the disease) could help understand how it improperly folds and aggregates into toxic plaques, Ishii said.New insights into Alzheimer’s, other neurodegenerative diseasesUnlike amyloid beta-42, amyloid beta-40 lacks the amino acid that carries the negative charge needed to form the salt bridge. Ishii added that this explains why the two proteins do not interact with one another, and that the findings provide new insight into amyloid propagation in Alzheimer’s, and possibly other neurodegenerative conditions as well.“Increasing evidence has suggested that formation and propagation of misfolded aggregates of 42-residue human amyloid β (Aβ(1–42), rather than of the more abundant Aβ(1–40), provokes the Alzheimer’s disease cascade,” the authors explained in their paper. “However, structural details of misfolded Aβ(1–42) have remained elusive. Here we present the atomic model of an Aβ(1–42) amyloid fibril, from solid-state NMR (ssNMR) data.”That atomic model “displays triple parallel-β-sheet segments that differ from reported structures of Aβ(1–40) fibrils” and is “incompatible with the triple-β-motif, because seeding with Aβ(1–42) fibrils does not promote conversion of monomeric Aβ(1–40) into fibrils via cross-replication,” they added. Their findings “provide insight into the Aβ(1–42)-selective self-replicating amyloid-propagation machinery in early-stage Alzheimer’s disease.”As Ishii said in a statement, drugs designed to act against amyloid beta-40 “may not work well against the more toxic amyloid beta-42.”Source: http://bit.ly/1JKek52